Downing Street insists UK will get 'first access' to Oxford coronavirus vaccine amid Donald Trump 'fast-tracking' claim

Downing Street has insisted the UK will get "first access" to a coronavirus vaccine developed by Oxford University amid reports that Donald Trump is considering fast-tracking its approval.
Oxford University's vaccine, currently being developed with pharmaceutical company AstraZeneca, is considered one of the leading candidates among more than 100 teams vying to produce one.
The White House is considering granting emergency authorisation for the vaccine in October, the Financial Times reported, citing sources familiar with the plan.
President Trump is lagging behind Democratic candidate Joe Biden in the polls, and getting a vaccine approved could be a way of boosting his numbers ahead of the presidential election in November.
A Number 10 spokesman said the UK Government has struck a deal with AstraZeneca to get “first access” to it once approved.
“We have been clear that we will only roll out a vaccine once it is deemed safe and effective by our regulators," said the Downing Street spokesman.
“AstraZeneca have entered into a number of agreements with other countries, they have the global licensing agreement with Oxford, but we have been clear: once it has been found to be effective, we have signed a deal for 100 million doses which means that once it is effective, the UK will get first access.”
The US has the highest number of deaths related to coronavirus and the most cases out of any country, according to Johns Hopkins University.
Almost 177,000 people are confirmed to have died with the virus, while more than 5.7 million have been infected.
The White House declined to comment on the claims. AstraZeneca said that it had not discussed the possibility of fast-tracking approval with the US authorities, adding: "It would be premature to speculate on that possibility."
Commenting on the prospect of the vaccine being fast-tracked, England’s deputy chief medical officer Dr Jenny Harries
told Sky News: “We have a global crisis… It is really important that everyone around the world has fair and safe access to vaccine development.
“Obviously those countries which are more developed have the facilities to develop the vaccine and get it safely out to their populations. But I think all public health colleagues would be wanting fair distribution.”
It comes after US officials announced plans to allow emergency use of blood plasma treatment for coronavirus patients.
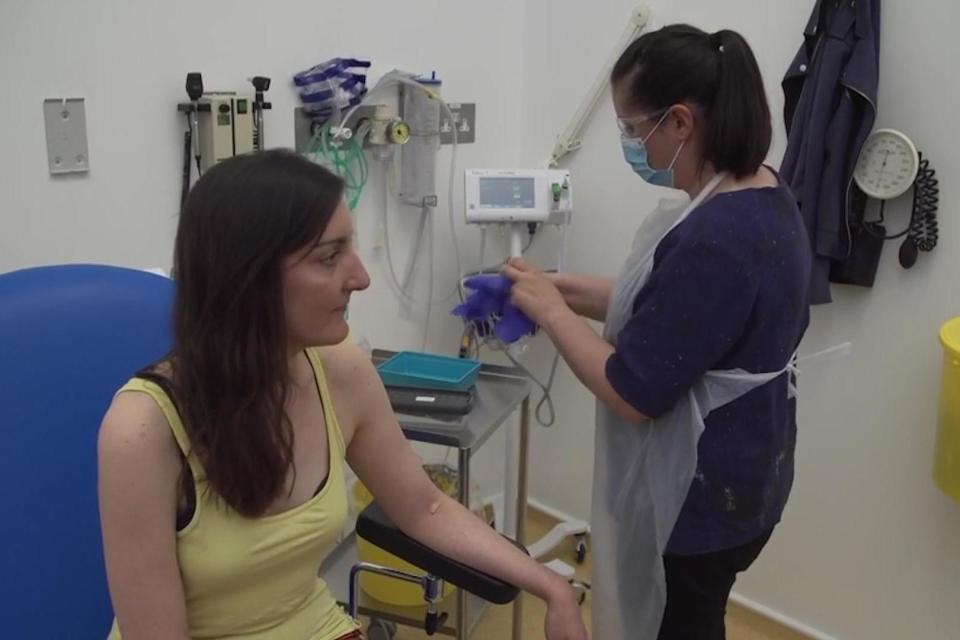
The study, led by the Mayo Clinic, indicated that patients treated within three days of diagnosis with convalescent plasma, which has high levels of antibodies, had an improved chance of survival.
NHS Blood and Transplant (NHSBT), which is supporting the British convalescent plasma research, said that randomised controlled trials are the “gold standard” and the ones published in the US are observational studies.
Research in the UK is assessing whether convalescent plasma donations can be transfused into patients who are struggling to develop their own immune response.
Additional reporting by PA Media.
Read more
Twitter puts notice on Trump tweet over 'misleading health claims'
PM says it's 'vitally important' children return to school- LIVE

 Yahoo Sport
Yahoo Sport 





































